- Home
- Products
- Prescription: drugs for allergy
- Dexamethasone-Darnitsa
Dexamethasone-Darnitsa
Corticosteroids for systemic use. Glucocorticoids. Dexamethasone- Solution for injections
- Eye drops
Dexamethasone should be administered intravenously or intramuscularly in urgent cases and when oral administration is impossible in:
- Diseases of the endocrine system
- Rheumatic diseases (as an adjuvant therapy in the period when the main therapy did not work, that is, in patients in whom the analgesic and anti-inflammatory effects of non-steroidal anti-inflammatory drugs were unsatisfactory)
- Skin diseases
- Allergic diseases (not curable by traditional treatment)
- Eye diseases
- Gastrointestinal diseases
- Diseases of the respiratory tract
- Hematological diseases
- Kidney diseases
- Malignant oncological diseases
- Edema of the brain
- Shock
- COVID-19
Main properties
- Release formInjectable solution
- ChildrenWith caution
- PregnantWith caution
- NursingContraindicated
- Release categoryPrescription medication
- AllergicsAllowed
- DiabeticsWith caution
- Temperature storage conditionsNot higher than 15 °С
Instructions for medical use
ЗАТВЕРДЖЕНО
Наказ Міністерства охорони
здоровʼя України
21.03.2019 № 629
Реєстраційне посвідчення
№ UA/0992/02/01
ІНСТРУКЦІЯ
для медичного застосування лікарського засобу
ДЕКСАМЕТАЗОН–ДАРНИЦЯ (DEXAMETHASONE–DARNITSA)
Склад:
діюча речовина: dexamethasone;
1 мл розчину містить дексаметазону натрію фосфату 4 мг;
допоміжні речовини: динатрію фосфат додекагідрат, калію дигідрофосфат, гліцерин, спирт бензиловий, динатрію едетат (трилон Б), вода для ін’єкцій.
Фармакотерапевтична група. Кортикостероїди для системного застосування. Глюкортикоїди. Дексаметазон. Код АТХ Н02А В02.
Фармакологічні властивості.
Фармакодинаміка.
Дексаметазон-Дарниця − синтетичний гормон кори надниркових залоз (кортикостероїд), який чинить глюкокортикоїдну дію. Чинить протизапальну та імуносупресивну дію, а також впливає на енергетичний метаболізм, обмін глюкози і (через негативний зворотний зв’язок) на секрецію фактора активації гіпоталамуса і трофічного гормону аденогіпофіза.
Механізм дії глюкокортикостероїдів досі не повністю з’ясований. Зараз отримана достатня кількість даних про механізм дії глюкокортикостероїдів на підтвердження того, що вони діють на клітинному рівні. У цитоплазмі клітин існують дві добре вивчені системи рецепторів. Через зв’язування з рецепторами глюкокортикоїдів глюкокортикостероїди чинять протизапальну та імуносупресивну дію і регулюють обмін глюкози, а внаслідок зв’язування з рецепторами мінералокортикоїдів вони регулюють метаболізм натрію, калію та водно-електролітну рівновагу.
Глюкокортикостероїди розчиняються у ліпідах і легко проникають у цільові клітини через клітинну мембрану. Зв’язування гормону з рецептором призводить до зміни конформації рецептора, що сприяє збільшенню його спорідненості з ДНК. Комплекс гормон/рецептор потрапляє у ядро клітини і зв’язується з регулюючим центром молекули ДНК, який також називають елементом глюкокортикоїдної відповіді (GRE). Активований рецептор, зв’язаний з GRE або зі специфічними генами, регулює транскрипцію м-РНК, що може бути збільшеною або зменшеною. Новоутворена м-РНК транспортується до рибосоми, після чого відбувається утворення нових білків. Залежно від цільових клітин і процесів, що відбуваються у клітинах, синтез білків може бути посилений (наприклад, утворення тирозинтрансамінази у клітинах печінки) або зменшений (наприклад, утворення IL-2 у лімфоцитах). Оскільки рецептори глюкокортикостероїдів є в усіх типах тканин, можна вважати, що глюкокортикостероїди діють на більшість клітин організму.
Клінічна ефективність та безпека - COVID-19
Клінічна ефективність
Індивідуально рандомізоване, контрольоване, відкрите, адаптивної платформи дослідження RECOVERY (Randomised Evaluation of Covid-19 Therapy)1, що ініційоване дослідником для оцінки наслідків потенційних методів лікування у пацієнтів, госпіталізованих із COVID-19.
Випробування проводили у 176 лікарнях Великобританії. 6425 пацієнтів було рандомізовано для отримання дексаметазону (2104 пацієнтів) або звичайного лікування (4321 пацієнтів). 89 % пацієнтів мали лабораторно підтверджену SARS-CoV-2 інфекцію.
При рандомізації 16 % пацієнтів отримували інвазивну штучну вентиляцію легень (ШВЛ) або екстракорпоральну мембранну оксигенацію, 60 % отримували лише кисень (з або без неінвазивної вентиляції), а 24 % не отримували ні того, ні іншого.
Середній вік пацієнтів становив 66,1 ± 15,7 року. 36 % пацієнтів були жінками. У 24 % пацієнтів в анамнезі був діабет, 27 % − із захворюванням серця та 21 % − із хронічними захворюваннями легенів.
Первинна кінцева точка
Летальність на 28 добу була значно нижчою у групі дексаметазону, ніж у групі звичайного лікування, де летальність зареєстрована у 482 з 2104 пацієнтів (22,9 %) та у 1110 з 4321 пацієнтів (25,7 %) відповідно (коефіцієнт частоти, 0,83; 95 % довірчий інтервал (ДІ), 0,75−0,93; Р < 0,001).
У групі дексаметазону частота летальних наслідків серед пацієнтів була нижчою, ніж у групі звичайного лікування, які отримують інвазивну ШВЛ (29,3 % проти 41,4 %; коефіцієнт частоти, 0,64; 95 % ДІ, 0,51−0,81) та у тих, хто отримує додатковий кисень без інвазивної ШВЛ (23,3 % проти 26,2 %; коефіцієнт частоти 0,82; 95 % ДІ від 0,72−0,94).
Не було чіткого ефекту дексаметазону серед пацієнтів, які не отримували жодних методів респіраторної підтримки при рандомізації (17,8 % проти 14,0 %; коефіцієнт частоти, 1,19; 95 % ДІ, 0,91−1,55).
Вторинна кінцева точка
У пацієнтів групи дексаметазону тривалість госпіталізації була меншою, ніж у групі звичайного лікування (медіана, 12 днів проти 13 днів) і більша ймовірність виписки з лікарні протягом 28 днів (коефіцієнт частоти, 1,10; 95 % ДІ, 1,03−1,17).
Відповідно до первинної кінцевої точки, найбільший ефект щодо скорочення тривалості госпіталізації до 28 днів спостерігався серед пацієнтів, які отримували інвазивну ШВЛ при рандомізації (коефіцієнт частоти 1,48; 95 % ДІ 1,16, 1,90), наступний – серед пацієнтів, які отримували лише кисень (коефіцієнт частоти, 1,15; 95 % ДІ 1,06−1,24) та без сприятливого ефекту у пацієнтів, які не отримують кисень (коефіцієнт норми 0,96; 95 % ДІ 0,85−1,08).
Результат | Дексаметазон | Звичайне лікування | Коефіцієнт частоти ризику* |
(N = 2104) | (N = 4321) | (95 % ДІ) | |
Кількість/загальна кількість пацієнтів (%) | |||
Первинна кінцева точка | 482/2104 (22,9) | 1110/4321 (25,7) | 0,83 (0,75-0,93) |
| Летальність у 28 днів | |||
| Вторинна кінцева точка |
|
|
|
| Виписаний із лікарні протягом 28 днів | 1413/2104 (67,2) | 2745/4321 (63,5) | 1,10 (1,03-1,17) |
Інвазивна ШВЛ або летальний наслідок†: | 456/1780 (25,6) | 994/3638 (27,3) | 0,92 (0,84-1,01) |
| 102/1780 (5,7) | 285/3638 (7,8) | 0,77 (0,62-0,95) |
| 387/1780 (21,7) | 827/3638 (22,7) | 0,93 (0,84-1,03) |
* Співвідношення показників було скориговано для віку з урахуванням результатів 28-денної летальності та виписки з лікарні. Співвідношення ризиків скориговано з урахуванням віку щодо результату отримання інвазивної ШВЛ або летального наслідку та його складових;
† З цієї категорії виключаються пацієнти, які отримували інвазивну ШВЛ при рандомізації.
Безпечність
У ході дослідження було зафіксовано 4 серйозні побічні явища, пов’язані з досліджуваним лікуванням, а саме: 2 випадки розвитку гіперглікемії, 1 випадок стероїдного індукованого психозу та 1 випадок кровотечі верхніх відділів шлунково-кишкового тракту. Всі випадки врегульовані.
Аналіз підгруп
Ефекти призначення дексаметазону на 28-денну смертність, залежно від віку та способу підтримки дихання, отримані при рандомізації2
Дексаметазон | Звичайний догляд | RR (95% Cl) | ||||||||
Відсутність кисню (x | 2 | = 0.70; p=0.40) | ||||||||
| 1 | ||||||||||
| < 70 | 10/197 (5.1%) | 18/462 (3.9%) | 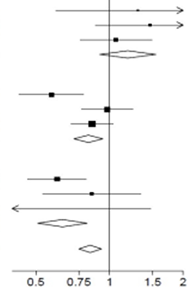
| 1.31 (0.60-2.83) | ||||||
| ≥ 70 < 80 | 25/114 (21.9%) | 35/224 (15.6%) | 1.46 (0.88-2.45) | |||||||
| ≥ 80 | 54/190 (28.4%) | 92/348 (26.4%) | 1.06 (0.76-1.49) | |||||||
| Проміжна сума | 89/501 (17.8%) | 145/1034 (14.0%) | 1.19 (0.91-1.55) | |||||||
| Тільки кисень (x | 2 | = 2.54; p=0.11) | ||||||||
| 1 | ||||||||||
| < 70 | 53/675 (7.9%) | 193/1473 (13.1%) | 0.58 (0.43-0.78) | |||||||
| ≥ 70 < 80 | 104/306 (34.0%) | 178/531 (33.5%) | 0.98 (0.77-1.25) | |||||||
| ≥ 80 | 141/298 (47.3%) | 311/600 (51.8%) | 0.85 (0.70-1.04) | |||||||
| Проміжна сума | 298/1279 (23.3%) | 682/2604 (26.2%) | 0.82 (0.72-0.94) | |||||||
| Механічна вентиляція (x | 2 | = 0.28; p=0.60) | ||||||||
| 1 | ||||||||||
| < 70 | 66/269 (24.5) % | 217/569 (38.1%) | 0.61 (0.46-0.81) | |||||||
| ≥ 70 < 80 | 26/49 (53.1%) | 58/104 (55.8%) | 0.85 (0.53-1.34) | |||||||
| ≥ 80 | 3/6 (50.0%) | 8/10 (80.0%) | 0.39 (0.10-1.47) | |||||||
| Проміжна сума | 95/324 (29.3%) | 283/683 (41.4%) | 0.64 (0.51-0.81) | |||||||
| ||||||||||
| Усі учасники | 482/2104 (22.9%) | 1110/4321 (25.7%) | 0.83 (0.75-0.93) р < 0.001 | |||||||
Дексаметазон краще | Звичайний догляд краще | |||||||||
Ефекти призначення дексаметазону на 28-денну смертність, залежно від способу підтримки дихання, отримані при рандомізації, та за наявності будь-якого хронічного захворювання3
Дексаметазон | Звичайний догляд | RR (95% Cl) | |||||||||
Відсутність кисню (x | 2 | = 0.08; p=0.78) | 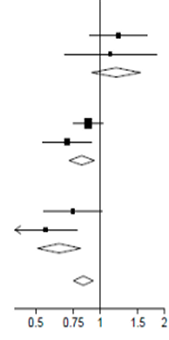
| ||||||||
1 | |||||||||||
| Попереднє захворювання | 65/313 (20.8%) | 100/598 (16.7%) | 1.22 (0.89-1.66) | ||||||||
Без попереднього захворювання | 24/188 (12.8%) | 45/436 (10.3%) | 1.12 (0.68-1.83) | ||||||||
| Проміжна сума | 89/501 (17.8%) | 145/1034 (14.0%) | 1.19 (0.91-1.55) | ||||||||
| Тільки кисень (x | 2 | = 2.05; p=0.15 |
| ||||||||
1 | |||||||||||
| Попереднє захворювання | 221/702 (31.5%) | 481/1473 (32.7%) | 0.88 (0.75-1.03) | ||||||||
Без попереднього захворювання | 77/577 (13.3%) | 201/1131 (17.8%) | 0.70 (0 54-0 91) | ||||||||
| Проміжна сума | 298/1279 (23.3%) | 682/2604 (26.2%) | 0.82 (0.72-0.94) | ||||||||
| Механічна вентиляція (x | 2 | = 1.52; p=0.22 |
| ||||||||
| 1 | |||||||||||
| Попереднє захворювання | 51/159 (32.1%) | 150/346 (43.4%) | 0.75 (0.54-1.02) | ||||||||
Без попереднього захворювання | 44/165 (26.7%) | 133/337 (39.5%) | 0.56 (0.40-0.78) | ||||||||
| Проміжна сума | 95/324 (29.3%) | 283/683 (41.4%) | 0.64 (0.51-0.81) | ||||||||
| |||||||||||
| Усі учасники | 482/2104(22.9%) | 1110/4321 (25.7%) | 0.83 (0.75-0.93) р < 0.001 | ||||||||
Дексаметазон краще | Звичайний догляд краще | ||||||||||
1 www.recoverytrial.net
2, 3 (джерело: Horby P. et al., 2020; https://www.medrxiv.org/content/10.1101/2020.06.22.20137273v1;
doi: https://doi.org/10.1101/2020.06.22.20137273).
Фармакокінетика.
Абсорбція.
Дексаметазон швидко абсорбується з місця ін’єкції. Максимальна концентрація у плазмі крові досягається протягом перших 5 хвилин при внутрішньовенному введенні та протягом 1 години при внутрішньом’язовому введенні.
Після місцевого ведення у суглоб або м’які тканини (вогнище запалення) всмоктування відбувається повільніше, ніж у випадку ін’єкційного введення.
При внутрішньовенному введенні початок дії є миттєвим; при внутрішньом’язовому введенні – через 8 годин.
Лікарський засіб належить до довгостроково діючих глюкокортикостероїдів. Дія триває 17–28 днів після внутрішньом’язового введення та від 3 днів до 3 тижнів після введення місцево.
Розподіл.
У плазмі крові приблизно 77 % дексаметазону зв’язуються з білками плазми, переважно з альбуміном. Лише невелика кількість дексаметазону зв’язується з іншими білками. Дексаметазон є жиророзчинним, тому він вільно проникає у клітини та міжклітинний простір. У центральній нервовій системі (гіпоталамус, гіпофіз) він зв’язується та діє через мембранні рецептори. У периферичних тканинах зв’язується та діє за допомогою цитоплазматичних рецепторів.
Біотрансформація.
Метаболізм дексаметазону відбувається у місці дії, тобто у самій клітині. Дексаметазон у першу чергу метаболізується у печінці, також, можливо, у нирках та інших тканинах.
Виведення.
Біологічний період напіввиведення дексаметазону становить 24–72 години. Близько 80 % введеного дексаметазону елімінується нирками у вигляді глюкуроніду протягом 24 годин.
Клінічні характеристики.
Показання.
Дексаметазон вводити внутрішньовенно або внутрішньом’язово у невідкладних випадках та при неможливості перорального застосування.
Захворювання ендокринної системи:
замісна терапія первинної або вторинної (гіпофізарної) недостатності надниркових залоз (крім гострої недостатності надниркових залоз, при якій гідрокортизон або кортизон є більш придатними, зважаючи на їх більш виражений гормональний ефект);
гостра недостатність надниркових залоз (гідрокортизон або кортизон є лікарськими засобами вибору; може бути необхідним сумісне застосування з мінералокортикоїдами, особливо при застосуванні синтетичних аналогів);
перед операціями та у випадках серйозних травм або захворювань у пацієнтів зі встановленою недостатністю надниркових залоз або при невизначеному адренокортикальному запасі;
шок, стійкий до традиційної терапії, при наявній або підозрюваній недостатності надниркових залоз;
уроджена гіперплазія надниркових залоз;
негнійне запалення щитовидної залози та тяжкі форми радіаційних тиреоїдитів.
Ревматологічні захворювання (як допоміжна терапія у період, коли базова терапія не подіяла, тобто у пацієнтів, у яких знеболювальна та протизапальна дія нестероїдних протизапальних засобів були незадовільними):
ревматоїдний артрит, включаючи ювенільний ревматоїдний артрит та позасуглобові прояви ревматоїдного артриту (ревматичні легені, зміни серця, очей, шкірний васкуліт);
синовіїт при остеоартрозі; посттравматичний остеоартроз; епікондиліт; гострий неспецифічний тендосиновіїт; гострий подагричний артрит; псоріатичний артрит; анкілозуючий спондиліт; системні захворювання сполучної тканини; васкуліт.
Захворювання шкіри:
пухирчатка; тяжка мультиформна еритема (синдром Стівенса-Джонсона); ексфоліативний дерматит; бульозний герпетиформний дерматит; тяжкі форми ексудативної еритеми; вузликова еритема; тяжкі форми себорейного дерматиту; тяжкі форми псоріазу; кропив’янка, що не піддається стандартному лікуванню; фунгоїдний мікоз; дерматоміозити.
Алергічні захворювання (що не піддаються традиційному лікуванню):
бронхіальна астма; контактний дерматит; атопічний дерматит; сироваткова хвороба; хронічний або сезонний алергічний риніт; алергія на ліки; кропив’янка після переливання крові.
Захворювання органів зору:
запальні захворювання очей (гострий центральний хоріоїдит, неврит зорового нерва); алергічні захворювання (кон’юнктивіти, увеїти, склерити, кератити, ірити); системні імунні захворювання (саркоїдоз, скроневий артеріїт); проліферативні зміни в очній ямці (ендокринна офтальмопатія, псевдопухлина); імуносупресорна терапія при пересадці рогівки. Розчин можливо вводити системно або ж місцево (введення під кон’юнктиву та ретробульбарне або парабульбарне введення).
Шлунково-кишкові захворювання:
для виведення пацієнта із критичного періоду при: виразковому коліті (тяжкий розвиток), хворобі Крона (тяжкий розвиток); хронічні аутоімунні гепатити; реакція відторгнення при пересадці печінки.
Захворювання дихальних шляхів:
симптоматичний саркоїдоз (симптоматично); гострий токсичний бронхіоліт; хронічний бронхіт та астма (при загостренні); вогнищевий або дисемінований туберкульоз легенів (разом з відповідною протитуберкульозною терапією); бериліоз (гранулематозне запалення); радіаційний або аспіраційний пневмоніт.
Гематологічні захворювання:
набута або вроджена хронічна апластична анемія; аутоімунна гемолітична анемія;
вторинна тромбоцитопенія у дорослих; еритробластопенія; гостра лімфобластомна лейкемія (індукційна терапія); ідіопатична тромбоцитопенічна пурпура у дорослих (лише внутрішньовенне введення – внутрішньом’язове введення протипоказане).
Ниркові захворювання:
імуносупресорна терапія при трансплантації нирки; стимулювання діурезу або зменшення протеїнурії при ідіопатичному нефротичному синдромі (без уремії) і порушення функції нирок при системному червоному вовчаку.
Злоякісні онкологічні захворювання:
паліативне лікування лейкемії та лімфоми у дорослих; гостра лейкемія у дітей; гіперкальціємія при злоякісних захворюваннях.
Набряк мозку:
набряк головного мозку внаслідок первинної чи метастатичної пухлини головного мозку, трепанація черепа та черепно-мозкові травми.
Шок:
шок, який не піддається класичному лікуванню; шок у пацієнтів із недостатністю кори надниркової залози; анафілактичний шок (внутрішньовенно після призначення адреналіну); перед операцією для запобігання шоку при підозрі або при встановленій недостатності кори надниркових залоз.
Коронавірусна хвороба 2019 (COVID-19):
- лікування коронавірусної хвороби 2019 (COVID-19) у дорослих та пацієнтів підліткового віку (віком від 12 років з масою тіла не менше 40 кг), яким потрібна додаткова киснева терапія.
Інші показання:
- туберкульозний менінгіт із субарахноїдальною блокадою (разом із належною протитуберкульозною терапією); трихінельоз із неврологічними симптомами або трихінельоз міокарда; кістозна пухлина апоневрозу або сухожилля (ганглія);
Показання для внутрішньосуглобового введення або введення у м’які тканини:
ревматоїдний артрит (тяжке запалення окремого суглоба); анкілозуючий спондиліт (коли запалені суглоби не піддаються традиційному лікуванню); псоріатичний артрит (олігоартикулярна форма та тендовагініт); моноартрит (після евакуації синовіальної рідини); остеоартрит суглобів (тільки у випадку синовіту та ексудації); позасуглобовий ревматизм (епікондиліт, тендовагініт, бурсит); гострий та подагричний артрит.
Місцеве введення (введення у місце ураження):
келоїдні ураження; гіпертрофічні, запальні та інфільтровані ураження при лишаї, псоріазі, кільцеподібній гранульомі, склерозуючому фолікуліті, дискоїдному вовчаку та шкірному саркоїдозі; дисковий червоний вовчий лишай; хвороба Урбаха-Оппенгейма; локалізована алопеція.
Протипоказання.
Підвищена чутливість до діючої речовини або до інших компонентів лікарського засобу.
Гострі вірусні, бактеріальні або системні грибкові інфекції (якщо не застосовується належна терапія).
Вакцинація живою вакциною.
Синдром Іценка-Кушинга.
Внутрішньом’язове введення протипоказане пацієнтам з тяжкими захворюваннями згортання крові.
У разі місцевого застосування – бактеріємія, системні грибкові інфекції, інфекції у місці застосування, у тому числі септичний артрит внаслідок гонореї чи туберкульозу, застосування пацієнтам із нестабільними суглобами.
Взаємодія з іншими лікарськими засобами та інші види взаємодій.
При одночасному застосуванні лікарського засобу з іншими лікарськими засобами можливі такі взаємодії:
з лікарськими засобами, що інгібують фермент CYP 3A4 (наприклад, кетоконазол, антибіотики-макроліди) – збільшення концентрації дексаметазону у плазмі крові; кетоконазол може пригнічувати наднирковий синтез глюкокортикостероїдів, таким чином, унаслідок зниження концентрації дексаметазону може розвиватися недостатність надниркових залоз;
з аміноглутетимідом, ефедрином, інгібіторами функції кори надниркових залоз (наприклад, мітотан), карбамазепіном, примідоном, рифампіцином, рифабутином, фенобарбіталом, фенітоїном – зменшення ефективності дексаметазону; при одночасному застосуванні лікарських засобів слід збільшити дозу дексаметазону;
з азатіоприном, антипсихотиками, іншими глюкокортикостероїдами, карбутамідом – підвищення ризику розвитку катаракти;
з альбендазолом, гепарином, калійуретиками, циклоспорином – збільшення ефективності останніх; при одночасному застосуванні циклоспорину та глюкокортикостероїдів можливе виникнення судом;
з амфотерицином В, β2-адреноміметиками, лікарськими засобами, що виводять калій з організму (наприклад, діуретики) – підвищення ризику розвитку гіпокаліємії, що може призводити до серцевої недостатності; при одночасному застосуванні амфотерицину В та глюкокортикостероїдів також збільшується ризик розвитку остеопорозу;
з антигіпертензивними засобами, натрійуретиками, празиквантелем, гіпоглікемічними засобами, саліцилатами, соматотропіном (у високих дозах) – зменшення ефективності останніх; при одночасному застосуванні дексаметазону та саліцилатів слід з обережністю знижувати дозу дексаметазону, оскільки при цьому може спостерігатися підвищення концентрації саліцилатів у плазмі крові та інтоксикація;
з антигістамінними засобами, м-холіноблокаторами, нітратами, трициклічними антидепресантами – ризик підвищення внутрішньоочного тиску; при одночасному застосуванні трициклічних антидепресантів та глюкокортикостероїдів також підвищується ризик депресії;
з антихолінестеразними засобами – підвищення ризику розвитку вираженої слабкості у пацієнтів із міастенією гравіс;
з вітаміном D – послаблення впливу останнього на всмоктування кальцію з кишечнику;
з гормонами щитовидної залози – підвищення кліренсу глюкокортикостероїдів;
з ізоніазидом, мексилетином – зниження концентрації останніх у плазмі крові внаслідок посилення їх метаболізму;
з імунодепресантами – підвищення ризику розвитку інфекцій, лімфоми або інших лімфопроліферативних порушень, пов’язаних з вірусом Епштейна-Барра;
з інгібіторами карбоангідрази – збільшення ризику розвитку остеопорозу;
з індометацином – посилення токсичних ефектів дексаметазону (внаслідок його витіснення зі зв’язку з альбумінами);
з живими противірусними вакцинами та на тлі інших видів імунізації – посилення ризику активації вірусів і розвитку інфекцій;
з кумариновими антикоагулянтами – зміна дії останніх; при одночасному застосуванні лікарських засобів слід провести моніторинг протромбінового часу;
з міорелаксантами – посилення вираженості і тривалості м’язової блокади на тлі гіпокаліємії, зумовленої глюкокортикостероїдами;
з нестероїдними протизапальними засобами, етанолом – підвищення ризику шлунково-кишкової кровотечі та утворення виразок;
з парацетамолом – посилення ризику розвитку гепатотоксичної дії парацетамолу внаслідок індукції «печінкових» ферментів і утворення його токсичного метаболіту;
з препаратами, що метаболізуються CYP 3A4 (індинавір, еритроміцин) – зниження концентрації останніх внаслідок збільшення їх кліренсу;
з ритордином – можливий розвиток набряку легенів; повідомлялося про летальний наслідок для породіллі через розвиток такого стану; одночасне застосування ритордину і дексаметазону протипоказане під час пологів;
із серцевими глікозидами – ризик порушення ритму серця у пацієнтів з гіпокаліємією та посилення токсичних ефектів глюкокортикостероїдів;
зі стероїдними гормональними лікарськими засобами (наприклад, андрогени, анаболіки, естрогени, пероральні контрацептиви) – поява вугрів та гірсутизму; естрогени, пероральні контрацептиви посилюють терапевтичні та токсичні ефекти глюкокортикостероїдів, знижуючи кліренс глюкокортикостероїдів;
з талідомідом – підвищення ризику розвитку токсичного епідермального некролізу.
Ергокальциферол та паратгормон перешкоджають розвитку остеопатії, спричиненої глюкокортикостероїдами.
Види взаємодії, що мають терапевтичні переваги: одночасне призначення дексаметазону і метоклопраміду, дифенгідраміду, прохлорперазину або антагоністів рецепторів 5-НТ3 (рецепторів серотоніну або 5-гідрокситриптаміну, тип 3, таких як ондансетрон або гранісетрон) ефективне для профілактики нудоти і блювання, спричинених хіміотерапією цисплатином, циклофосфамідом, метотрексатом, фторурацилом.
Взаємодія з дексаметазоном та усіма вищезгаданими лікарськими засобами може спотворити тест пригнічення впливу дексаметазону. Це слід враховувати при оцінці результатів тесту.
Антациди зменшують всмоктування дексаметазону у шлунку. Дія дексаметазону при одночасному прийомі з їжею та алкоголем не досліджена, проте одночасне вживання ліків та їжі з високим вмістом натрію не рекомендується.
Куріння не впливає на фармакокінетику дексаметазону.
Особливості застосування.
До початку і під час проведення терапії глюкокортикостероїдами необхідно проводити загальний аналіз крові, контролювати рівень глікемії та вміст електролітів у плазмі крові.
Під час лікування дексаметазоном (особливо тривалого) необхідний нагляд окуліста, контроль артеріального тиску і водно-електролітного балансу, зокрема рівня калію у сироватці крові, а також картини периферичної крові і рівня глікемії.
Під час парентерального лікування кортикостероїдами у поодиноких випадках можуть спостерігатися реакції гіперчутливості, тому треба вжити належних заходів до початку лікування дексаметазоном, зважаючи на можливість алергічних реакцій (особливо у пацієнтів з алергічними реакціями на будь-які інші ліки в анамнезі).
При відміні лікарського засобу у разі тривалого лікування можливий розвиток синдрому відміни (без видимих ознак недостатності надниркових залоз) з такими симптомами: підвищена температура, нежить, почервоніння кон’юнктиви, головний біль, запаморочення, сонливість або дратівливість, загальмованість, біль у м’язах і суглобах, нудота, блювання, зменшення маси тіла, загальна слабкість, судоми. У зв’язку з цим дозу дексаметазону слід зменшувати поступово. Раптове припинення прийому може призвести до летального наслідку.
Якщо пацієнт перебуває у стані тяжкого стресу (через травму, операцію або тяжке захворювання) під час терапії або під час припинення терапії дексаметазоном, дозу слід збільшити або застосовувати гідрокортизон чи кортизон.
Пацієнтам, які застосовували дексаметазон тривалий час і зазнають тяжкого стресу після припинення терапії, слід відновити прийом дексаметазону, оскільки спричинена недостатність надниркових залоз може продовжуватися протягом кількох місяців після припинення лікування.
Лікування дексаметазоном або природними глюкокортикостероїдами може приховати симптоми вже наявної або нової інфекції, а також симптоми кишкової перфорації. Під час лікування слід уникати контакту з хворими на застуду або інші інфекції.
Дексаметазон може спричинити загострення системної грибкової інфекції, латентний амебіаз і туберкульоз легень.
Пацієнти з туберкульозом легень в активній формі мають отримувати дексаметазон (разом із засобами проти туберкульозу) тільки при швидкоплинному або розсіяному туберкульозі легень. Пацієнти з неактивною формою туберкульозу легень, які лікуються дексаметазоном, або пацієнти, які реагують на туберкулін, мають отримувати хімічні профілактичні засоби.
Вакцинація живою вакциною протипоказана під час лікування дексаметазоном. Вакцинація неживою вірусною або бактеріальною вакциною не приводить до очікуваного розвитку антитіл і не дає очікуваного захисного ефекту. Дексаметазон не призначати за 8 тижнів до вакцинації і розпочинати застосовувати не раніше ніж через 2 тижні після вакцинації.
Лікарський засіб з обережністю призначати інфекційним хворим, особливо з вітряною віспою та кором, оскільки ці захворювання при застосуванні дексаметазону протікають у тяжчій формі. Тому особам, які не хворіли на ці захворювання, слід бути обережними, щоб максимально виключити інфікування. У випадку контакту з хворими необхідно одразу ж звернутися до лікаря. Рекомендовано профілактичне лікування імуноглобуліном.
Глюкокортикостероїди слід з обережністю призначати хворим на пухирчатий лишай очей (herpes sіmplex), тому що їхнє застосування може призвести до перфорації рогівки.
Обережність і медичний нагляд рекомендовані хворим на остеопороз, артеріальну гіпертензію, серцеву недостатність, туберкульоз, глаукому, печінкову або ниркову недостатність, цукровий діабет, гастрит, езофагіт, дивертикуліт, активну пептичну виразку, з нещодавнім кишковим анастомозом, хворим на коліт і епілепсію, тиреотоксикоз, ожиріння (III-IV ступеня), нефроуролітіаз, гіперліпідемію, поліомієліт (за винятком форми бульбарного енцефаліту), гіпоальбумінемію та хворим у станах, що призводять до її виникнення, хворим з імунодефіцитними станами (у т. ч. СНІД або ВІЛ-інфікування), лімфаденітом після щеплення БЦЖ.
Особливого догляду потребують пацієнти протягом перших тижнів після інфаркту міокарда, пацієнти з тромбоемболією, тяжкою міастенією гравіс, гіпотиреозом, психозом або психоневрозом, а також пацієнти літнього віку.
Під час лікування дексаметазоном може спостерігатися загострення цукрового діабету або перехід від латентної фази до клінічних проявів.
Дія глюкокортикостероїдів посилюється у хворих на цироз печінки або гіпотиреоз.
Хворі з порушенням водно-електролітного балансу повинні бути обережними при прийомі дексаметазону, тому що середні та великі дози глюкокортикостероїдів можуть викликати в організмі затримку солі та рідини, а також підвищену екскрецію калію. У даних випадках показане обмеження вживання солі та додатковий прийом калію. Усі кортикостероїди посилюють процес виведення кальцію, внаслідок чого може бути порушена секреція мінералокортикоїдів. Тому показане додаткове призначення солі та/або мінералокортикоїдів.
Тривалий прийом глюкокортикостероїдів може призвести до розвитку задньої субкапсулярної катаракти, глаукоми, з пошкодженням очного нерва, а також збільшує ризик вторинних вірусних або грибкових очних інфекцій.
Рекомендується бути обережними пацієнтам, які одужують після операції або перелому кісток, оскільки дексаметазон може уповільнити загоєння ран і утворення кісткової тканини.
Системні кортикостероїди не слід припиняти пацієнтам, які вже приймають системні (пероральні) кортикостероїди з інших причин (наприклад, пацієнти з хронічною обструктивною хворобою легенів) та не потребують додаткового кисню.
Особливої уваги потребує питання застосування системних глюкокортикостероїдів хворим з тяжкими афективними розладами, що включають депресивний, маніакально-депресивний психоз, попередній стероїдний психоз, у тому числі хворим з такими порушеннями в анамнезі. Пацієнтів та/або опікунів слід попередити щодо можливості розвитку серйозних побічних ефектів з боку психіки. Симптоми зазвичай проявляються протягом кількох днів або тижнів після початку лікування. Ризик виникнення даних побічних ефектів вищий при застосуванні високих доз. Більшість реакцій зникає після зменшення дози або відміни лікарського засобу, хоча інколи є необхідним специфічне лікування.
Потрібно спостерігати та вчасно виявляти зміни психічного стану, особливо депресивний настрій, суїцидальні думки та наміри. З особливою обережністю потрібно застосовувати кортикостероїди пацієнтам з афективними розладами, наявними в анамнезі, особливо пацієнтам з алергічними реакціями на будь-які інші ліки в анамнезі, а також у найближчих родичів. При розвитку подібних симптомів необхідно звернутися до лікаря. Також психічні розлади можуть спостерігатися при відміні глюкокортикоїдів.
Глюкокортикостероїди можуть спотворювати результати алергічних нашкірних тестів.
Повідомлялося про гіпертрофічну кардіоміопатію після системного застосування кортикостероїдів недоношеним немовлятам. Здебільшого це явище було оборотним після припинення лікування. Рекомендується проводити діагностичну оцінку і моніторинг функції та структури серця у недоношених дітей, які отримували дексаметазон для системного застосування.
Дітей можна лікувати дексаметазоном тільки у разі явної необхідності. Під час лікування дексаметазоном необхідний ретельний нагляд за ростом і розвитком дітей.
Є дані, які свідчать про довгострокові неврологічні побічні ефекти після раннього лікування (< 96 годин) недоношених дітей із хронічним захворюванням легенів при початкових дозах 0,25 мг/кг двічі на день.
Внутрішньосуглобове застосування лікарського засобу може призвести до місцевих або системних побічних реакцій. Часте застосування може спричинити ураження хряща або некроз кістки.
Перед внутрішньосуглобовим введенням дексаметазону слід видалити з суглоба синовіальну рідину та дослідити її (перевірити на наявність інфекції). Не застосовувати лікарський засіб пацієнтам з інфікованими суглобами. Якщо інфекція суглоба розвивається після ін’єкції, треба розпочати належну терапію антибіотиками.
Часті внутрішньосуглобові ін’єкції можуть травмувати тканини суглоба. Під час лікування пацієнтам слід уникати надмірних навантажень на ушкоджені суглоби до повного зникнення запального процесу, навіть при настанні симптоматичного поліпшення.
Слід припинити застосування лікарського засобу хворим, у яких при внутрішньосуглобовому введенні глюкокортикостероїдів значно посилився біль, що супроводжується припухлістю і подальшим обмеженням рухливості суглоба, гарячкою і загальним нездужанням (дані симптоми свідчать про виникнення септичного артриту). У випадку розвитку септичного артриту і при підтвердженні діагнозу сепсису необхідно призначити відповідну антибактеріальну терапію.
Феохромоцитомний криз
Повідомлялося про розвиток феохромоцитомного кризу, який може призвести до летального наслідку, після лікування кортикостероїдами для системного застосування. Кортикостероїди слід призначати пацієнтам з підозрою на феохромоцитому або з діагностованою феохромоцитомою лише після відповідної оцінки співвідношення користь/ризик.
Допоміжні речовини
Цей лікарський засіб містить менше 1 ммоль (23 мг)/дозу натрію, тобто практично вільний від натрію.
Застосування у період вагітності або годування груддю.
Період вагітності.
Глюкокортикостероїди проникають крізь плаценту і досягають високих концентрацій у плоді. Шкідливий ефект на плід та новонароджену дитину не може бути виключеним. Дексаметазон пригнічує внутрішньоутробний розвиток дитини. За деякими даними, навіть фармакологічні дози глюкокортикостероїдів підвищують ризик недостатності плаценти, олігогідрамніону, уповільненого розвитку плода або його внутрішньоматкової загибелі, підвищення кількості лейкоцитів (нейтрофілів) у плода і недостатності надниркових залоз. Відсутні дані щодо тератогенної дії дексаметазону. Дітей, народжених матерями, яким призначали глюкокортикостероїди у період вагітності, треба ретельно перевіряти на наявність недостатності надниркових залоз.
Також деякі дослідження показали підвищений ризик неонатальної гіпоглікемії після пренатального короткотривалого застосування кортикостероїдів жінкам із ризиком пізніх передчасних пологів.
Лікарський засіб застосовувати у невідкладних випадках, коли очікувана користь для матері перевищує потенційний ризик для плода.
Особлива обережність рекомендується при прееклампсії. Відповідно до загальних рекомендацій, при лікуванні у період вагітності глюкокортикостероїдами повинна бути використана найнижча дієва доза для контролю основного захворювання.
Жінкам, які застосовували глюкокортикостероїди у період вагітності, рекомендується застосовувати їх додаткові дози під час пологів. У випадку затяжних пологів або у разі планування кесаревого розтину рекомендується внутрішньовенне введення 100 мг гідрокортизону кожні 8 годин.
Період годування груддю.
Лікарський засіб протипоказаний у період годування груддю (за винятком невідкладних випадків).
Глюкокортикостероїди проникають у грудне молоко. У разі застосування дексаметазону, особливо у дозі понад фізіологічні норми (близько 1 мг), не рекомендується годування груддю, оскільки це може призвести до уповільнення росту дитини та зменшення секреції ендогенних кортикостероїдів.
Здатність впливати на швидкість реакції при керуванні автотранспортом або іншими механізмами.
Даних немає, однак слід враховувати можливість розвитку побічних реакцій з боку нервової системи та органів зору.
Спосіб застосування та дози.
Лікарський засіб застосовувати дорослим та дітям від народження.
Вводити внутрішньовенно (у вигляді ін’єкції або інфузії), внутрішньом’язово або місцево – за допомогою внутрішньосуглобових ін’єкцій чи ін’єкцій у місце ураження на шкірі або в інфільтрат м’яких тканин. Як розчинник для внутрішньовенної інфузії застосовувати 0,9 % розчин натрію хлориду або 5 % розчин глюкози.
При застосуванні немовлятам, особливо недоношеним, розчини, призначені для внутрішньовенного введення або подальшого розчинення лікарського засобу, не повинні містити консерванти.
При змішуванні лікарського засобу з розчинником для інфузії слід дотримуватись правил асептики. Суміш слід застосовувати протягом 24 годин, оскільки розчини для інфузій зазвичай не містять консервантів.
Лікарські засоби для парентерального введення слід візуально перевіряти на наявність сторонніх включень та зміни кольору кожного разу перед введенням.
Дозу лікарського засобу слід визначати індивідуально, відповідно до захворювання конкретного пацієнта, передбаченого періоду лікування, переносимості глюкокортикостероїдів і реакції організму.
Дорослим.
Лікарський засіб вводити парентерально у невідкладних випадках, коли пероральна терапія неможлива, та у випадках, зазначених у розділі «Показання».
Внутрішньовенне та внутрішньом’язове введення.
Лікарський засіб застосовувати у початковій дозі від 0,5 до 9 мг/добу, при необхідності дозу можна збільшити.
Початкові дози дексаметазону слід застосовувати до появи клінічної реакції, а потім дозу слід поступово зменшувати до найнижчої клінічно ефективної. У разі застосування високих доз протягом періоду, що перевищує кілька днів, дозу слід зменшувати поступово протягом кількох наступних днів або навіть протягом більш тривалого періоду.
Місцеве введення.
Зазвичай лікарський засіб застосовувати у дозі 0,2–4 мг, зокрема:
– великі суглоби (наприклад, колінні) – 2–4 мг;
– маленькі суглоби (наприклад, міжфалангові, скронево-нижньощелепні) – 0,8–1 мг;
– синовіальна сумка – 2–3 мг;
– сухожильні оболонки – 0,4–1 мг;
– інфільтрація м’яких тканин – 2–6 мг;
– ганглії – 1–2 мг.
Повторне введення в суглоб можливе після 3–4 місяців. Введення може бути виконано 3 або 4 рази в один суглоб протягом усього життя. Частіше внутрішньосуглобове введення може пошкодити суглобовий хрящ та спричинити кістковий некроз. Внутрішньосуглобові ін’єкції рекомендується робити не більше ніж у 2 суглоби одночасно.
Доза дексаметазону, що вводиться у місце пошкодження, прирівнюється до внутрішньосуглобової дози. Дексаметазон рекомендується вводити не більше ніж у 2 місця пошкодження одночасно.
Внутрішньосуглобові ін’єкції глюкокортикостероїдів можуть призвести до розвитку системних реакцій на додаток до місцевих.
Слід уникати внутрішньосуглобового введення глюкокортикостероїдів в інфіковані суглоби та нестабільні або деформовані суглоби.
Дітям.
Замісна терапія: лікарський засіб застосовувати у дозі 0,02 мг/кг маси тіла (0,67 мг/м2 площі поверхні тіла) на добу за 3 ін’єкції, або 0,008−0,01 мг/кг маси тіла (0,2–0,3 мг/м2 площі поверхні тіла).
Інші показання: лікарський засіб застосовувати у дозі 0,02–0,1 мг/кг (0,8–5 мг/м2 площі поверхні тіла) кожні 12–24 години.
Для лікування COVID-19
Дорослим та дітям (підліткам віком від 12 років і старше з масою тіла не менше 40 кг): по 6 мг дексаметазону внутрішньовенно 1 раз на добу курсом до 10 днів.
Тривалість лікування визначається індивідуально, відповідно до клінічного стану пацієнта.
Пацієнти літнього віку, із порушеннями функції нирок або печінки.
Коригування дози не потрібно.
Для порівняння нижче наведені еквівалентні дози різних глюкокортикостероїдів у міліграмах:
Дексаметазон 0,75 мг | Преднізон 5 мг |
Кортизон 25 мг | Метилпреднізолон 4 мг |
Гідрокортизон 20 мг | Тріамцинолон 4 мг |
Преднізолон 5 мг | Бетамезон 0,75 мг |
Діти.
Лікарський засіб застосовувати з періоду новонародженості тільки у разі крайньої необхідності. Під час лікування дексаметазоном необхідний ретельний нагляд за ростом і розвитком дітей та підлітків.
Передозування.
Симптоми. Є поодинокі повідомлення про гостре передозування або про летальний наслідок через гостре передозування. Передозування (зазвичай тільки після кількох тижнів застосування надмірних доз) проявляється симптомами посилення описаних побічних ефектів, зокрема: перш за все синдром Іценка-Кушинга, акне, екхімози, гірсутизм, нудота, анорексія, пептична виразка травного тракту, артралгія, міопатія, міалгія, задишка, запаморочення, втрата свідомості, гарячка, підвищення артеріального тиску, гіперліпідемія, остеопороз, ортостатична гіпотензія, розвиток інфекцій, стероїдний діабет, «місяцеподібне» обличчя, статева дисфункція.
Лікування: симптоматична та підтримуюча терапія. Специфічного антидоту немає. Гемодіаліз не ефективний.
Побічні реакції.
Частота появи побічних реакцій залежить від дози і тривалості лікування.
Побічні реакції при короткотривалому лікуванні.
З боку шлунково-кишкового тракту: пептична виразка, гострий панкреатит.
З боку ендокринної системи: тимчасове пригнічення функції надниркових залоз.
З боку обміну речовин, метаболізму: зниження толерантності до вуглеводів, збільшення апетиту, збільшення маси тіла, гіпертригліцеридемія.
З боку психіки: психічні розлади.
З боку імунної системи: реакції гіперчутливості.
Побічні реакції при довготривалому лікуванні.
З боку органів зору: катаракта, глаукома.
З боку ендокринної системи: тривале пригнічення функції надниркових залоз, затримка росту дітей, передчасне закриття епіфізарних зон росту.
З боку обміну речовин, метаболізму: ожиріння.
З боку серцево-судинної системи: артеріальна гіпертензія, телеангіектазія.
З боку імунної системи: зменшення імунної відповіді та підвищена сприйнятливість до інфекційних захворювань.
З боку шкіри та підшкірної клітковини: потоншання шкіри.
З боку опорно-рухової системи та сполучної тканини: м’язова атрофія, остеопороз, переломи трубчатих кісток, асептичний некроз кісток.
Побічні реакції, що можуть виникати в окремих органах та системах при лікуванні дексаметазоном.
З боку органів зору: підвищення внутрішньоочного тиску, глаукома, катаракта, екзофтальм, папілоедема, потоншання рогівки або склери, ретролетальна фіброплазія, загострення офтальмологічних вірусних або грибкових захворювань.
З боку респіраторної системи, органів грудної клітки та середостіння: набряк легенів.
З боку шлунково-кишкового тракту: нудота, блювання, диспепсія, гикавка, метеоризм, езофагіт, кандидоз стравоходу, підвищення або зниження апетиту; рідко – пептичні виразки шлунка та дванадцятипалої кишки, можливі також виразкові перфорації та кровотеча у травному тракті (криваве блювання, мелена), панкреатит і перфорація жовчного міхура та кишечнику (особливо у пацієнтів з хронічним запаленням кишечнику), атонія травного тракту.
З боку печінки і жовчовивідних шляхів: підвищення рівня ферментів печінки, гепатомегалія.
З боку ендокринної системи: пригнічення функції та атрофія надниркових залоз (зменшення реакції на стрес), пригнічення гіпоталамо-гіпофізарно-надниркової системи, синдром Іценка-Кушинга, затримка статевого розвитку у дітей, порушення секреції статевих гормонів (порушення менструального циклу, аменорея, гірсутизм, імпотенція), гіперглікемія, «стероїдний» цукровий діабет, перехід латентного діабету в клінічно активну форму, зменшення переносимості вуглеводів, підвищена потреба в інсуліні або пероральних протидіабетичних лікарських засобах у хворих на цукровий діабет.
З боку обміну речовин, метаболізму: підвищене виведення іонів кальцію, гіпокальціємія, підвищення маси тіла, негативний баланс азоту (підвищений розпад білків), затримка рідини та іонів натрію (периферичні набряки), гіпернатріємія, підвищене виділення іонів калію, гіпокаліємічний синдром: гіпокаліємія, гіпокаліємічний алкалоз, аритмія, міалгія або спазм м’язів, синдром лізису пухлини.
З боку нервової системи: після лікування можлива поява набряку диска зорового нерва і підвищення внутрішньочерепного тиску (псевдопухлина), загострення епілепсії, запаморочення, непритомність, судоми, головний біль, гіперкінезія, неврит, невропатія, парестезії; у дітей лікарський засіб може спричинити параліч кори головного мозку.
З боку психіки: зміни особистості та поведінки, які часто проявляються у вигляді афективних порушень (дратівливість, ейфорія, делірій, параноя, нервозність, тривожність, дезорієнтація, депресія, лабільність настрою, суїцидальні думки), порушення сну, пізнавальна дисфункція (включаючи сплутаність свідомості і амнезію), психотичні реакції (включаючи манію, галюцинації, психози та загострення шизофренії).
З боку серцево-судинної системи: пароксизмальна брадикардія, зупинка серця, серцеві аритмії, напад стенокардії, збільшення серця, судинна недостатність, застійна серцева недостатність, хронічна серцева недостатність, жирова емболія, артеріальна гіпертензія, гіпертрофічна кардіоміопатія у недоношених немовлят; дуже рідко – розрив серця у пацієнтів, які нещодавно перенесли інфаркт міокарда; також можливі політопна шлуночкова екстрасистола, гіпертонічна енцефалопатія, тахікардія, тромбоемболія, тромбофлебіт, васкуліт.
З боку крові та лімфатичної системи: випадки тромбоемболії, тромбофлебіт, зменшення кількості моноцитів і/або лімфоцитів, лейкоцитоз, еозинофілія (як і при застосуванні інших глюкокортикоїдів), рідко – тромбоцитопенія і нетромбоцитопенічна пурпура.
З боку імунної системи: реакції гіперчутливості, у тому числі висипання, свербіж, гіперемія, алергічний дерматит, кропив’янка, набряк Квінке, бронхоспазм, анафілактичний шок, імуносупресія.
З боку шкіри та підшкірної клітковини: пригнічення регенеративних та репаративних функцій шкіри, уповільнене загоєння ран, потоншена і чутлива шкіра, сухість шкіри, атрофія шкіри, гіпер- або гіпопігментація, порушення розподілу підшкірної клітковини, екхімоз, стерильний абсцес, петехії, вугри, стрії, телеангіектазія, пригнічення реакції на шкірні тести і щеплення.
З боку опорно-рухової системи та сполучної тканини: м’язова слабкість, атрофія м’язів, стероїдна міопатія (м’язова слабкість спричиняє м’язовий катаболізм), остеопороз (підвищене виведення кальцію), переломи трубчастих кісток або компресійні переломи хребта, асептичний остеонекроз (частіше – асептичний некроз головок кісток стегон і плечей), розриви сухожиль (особливо при одночасному застосуванні деяких хінолонів), затримка росту і процесу мінералізації кісток у дітей, передчасне закриття епіфізарних зон росту.
Загальні розлади та реакції у місці введення: реакції гіперчутливості, у тому числі висипання, свербіж, гіперемія і поколювання шкіри, безболісне руйнування суглоба, що симптоматично нагадує нейрогенну артропатію (суглоб Шарко).
Інші: незвична слабкість та підвищена втомлюваність, підвищення ризику виникнення або загострення грибкових, вірусних або бактеріальних інфекцій, розвиток умовно-патогенних інфекцій, пригнічення регенеративних і репаративних процесів, набряки, підвищене потовиділення, лейкоцитурія, синдром відміни. Лікарський засіб може викликати фізичну залежність.
Ознаки синдрому відміни глюкокортикостероїдів.
У пацієнтів, які тривалий час лікувалися дексаметазоном, під час дуже швидкого зниження дози може спостерігатися синдром відміни та виникати недостатність надниркових залоз, артеріальна гіпотензія, можливий летальний наслідок. У деяких випадках симптоми синдрому відміни можуть бути подібними до ознак погіршення або рецидиву захворювання, від якого пацієнт лікувався.
Термін придатності. 2 роки.
Умови зберігання.
Зберігати в оригінальній упаковці при температурі не вище 15 °С. Не заморожувати. Зберігати у недоступному для дітей місці.
Несумісність.
Одночасне застосування ритордину і дексаметазону протипоказано, оскільки це може призвести до набряку легенів. Повідомлялося про летальний наслідок для породіллі через розвиток такого стану.
Упаковка.
По 1 мл в ампулі; по 5 ампул у контурній чарунковій упаковці; по 2 контурні чарункові упаковки в пачці; по 5 або 10 ампул у контурній чарунковій упаковці; по 1 контурній чарунковій упаковці в пачці.
Категорія відпуску. За рецептом.
Виробник. ПрАТ «Фармацевтична фірма «Дарниця».
Місцезнаходження виробника та його адреса місця провадження діяльності.
Україна, 02093, м. Київ, вул. Бориспiльська, 13.
Дата останнього перегляду. 05.09.2024
